|
However the model used today is closest to the Bohr model of the atom, using the quantized shells to contain the electrons. In 1926, the Austrian physicist Erwin Schrödinger created a quantum mechanical model of the atom by combining the equations for the behavior of waves with the de Broglie equation to generate a mathematical model for the distribution of electrons in an atom. This was because it was impossible for the cloud of negative electrons proposed by Rutherford to exist, as the negative electrons would be drawn to the positive nucleus, and the atom would collapse in on itself. In 1913, Niels Bohr proposed a model of the atom where the electrons were contained within quantized shells that orbited the nucleus. Temperature is measured with a thermometer. This theory was then disproved by Ernest Rutherford and the gold foil experiment in 1911, where Rutherford shot alpha particles at gold foil, and noticed that some went through and some bounced back, implying the existence of a positive nucleus. Temperature is a physical quantity that expresses quantitatively the attribute of hotness or coldness. This model was known as the 'plum pudding' model. The first model of the atom was developed by JJ Thomson in 1904, who thought that atoms were composed purely of negatively charged electrons. This is not coming from an electron in an atom mind you. Physicists manage to detect electromagnetic radiation from an electron. This is an article about an electron moving in circular motion. In Rutherford's model, the atom would eventually cease to exist because the electron would collapse into the nucleus (something which we never observe). According to Maxwell, charges in circular motion must emit radiation. Next, the electron as proposed by Rutherford moves in circular orbit. Rutherford's model cannot explain this phenomenon. The problem is, atoms emit and absorb certain wavelengths of light which shows that the atom has different energy levels. Admirable but it is still a flawed model. Before this, J.J Thomson proposed his own atomic model: The atom is made out of a positive sphere with electrons in it. The electron loses energy, spirals into the. Too many photons are emitted in the UV region. What is that shortcoming Too many photons are emitted in the visible region of the emission spectrum. This may seem obvious (we see that in most elementary textbooks). The classical solar system model has a significant shortcoming. Describe this model of the Circle all correct answers) hydrogen. 
This is the final atomic model you discussed in chapter 2. Now click on and select next model, Rutherford's Classical Solar System model. Rutherford's Classical Solar System Model 11. 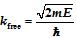
The electrons on the other hand, orbit this nucleus thus, completing the model of the atom. (You should let the simulation run for -1 minute with white light then take a snapshot & save it). As far as I know, Rutherford's atomic model says that atoms have a center (the nucleus) of concentrated positive charge and this center is very very small compared to the actual size of the atom.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
